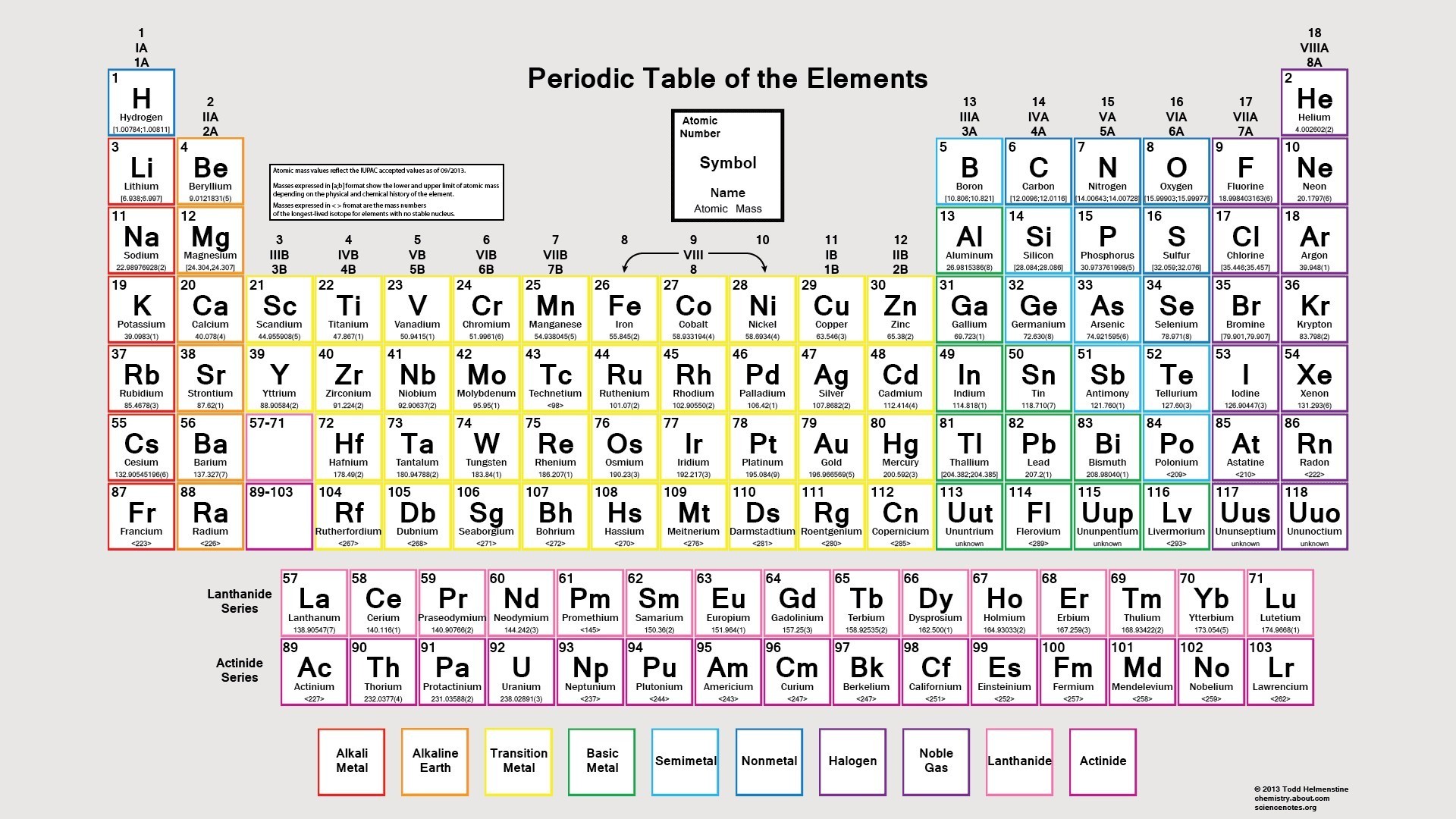
The lightest chemical element is Hydrogen and the heaviest is Hassium. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). This list contains the 118 elements of chemistry. Separation and Concentration Purification RequestĬhemical elements listed by atomic mass The elements of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects.Plant Inspection & Process Optimalisation.Molecules of the same type follow this trend.
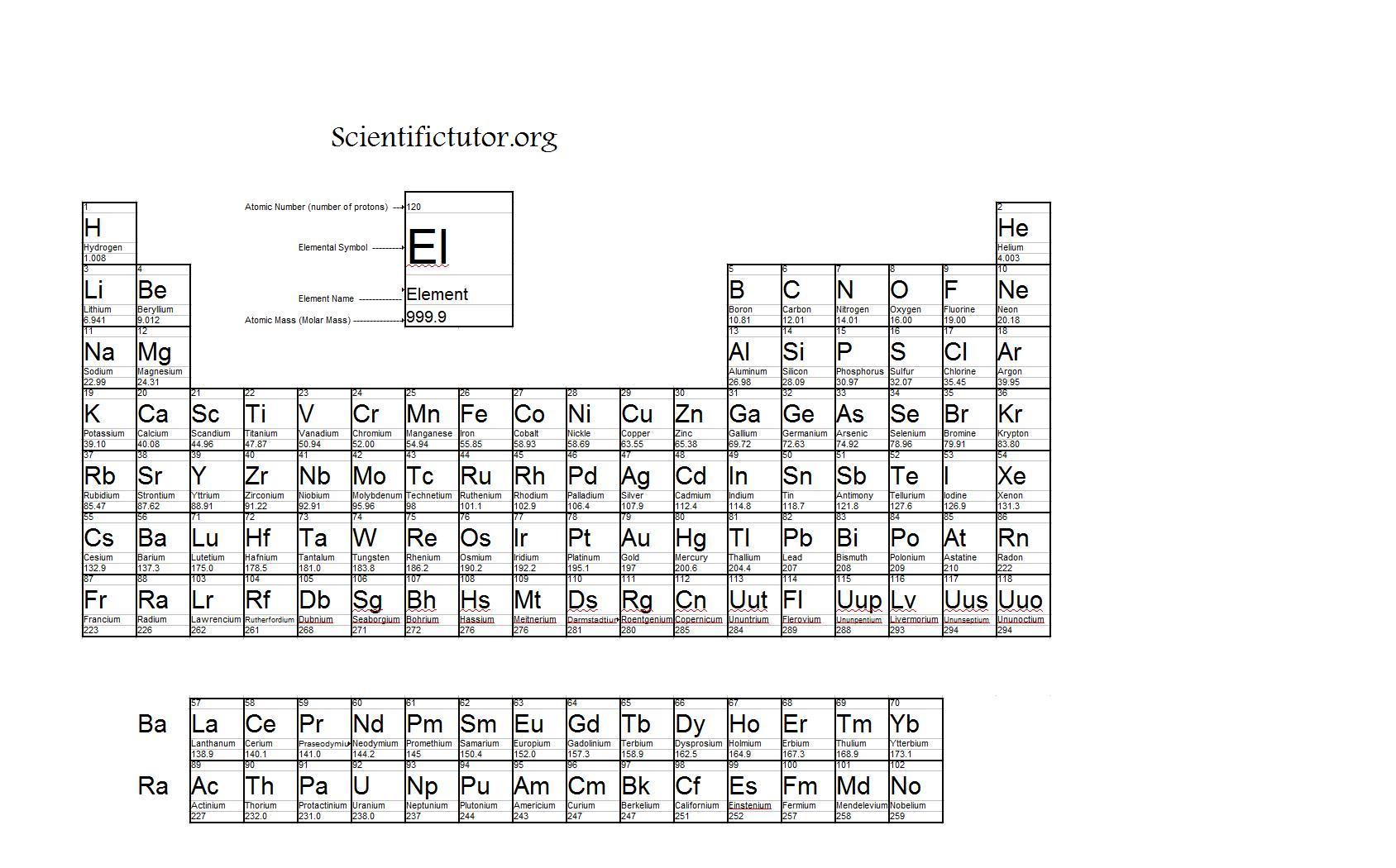
While the molecular weight is the sum of the atomic weights of the elements present in the molecule.įor elements, the greater the molar mass, the greater the boiling point. The molecular mass is the sum of the atomic masses of the elements present in the molecule. It is calculated by adding the number of protons and neutrons (electrons are considered too light and are negligible). The atomic mass is the mass of a single atom/isotope. The atomic weight of an element is an average of the weight of its isotopes with respect to their relative abundances. The molecular mass would then be: $$(2*1.01\frac$$ Water is made up of 2 hydrogens and 1 oxygen, and the atomic mass of hydrogen is 1.01 g/mol and the atomic mass of oxygen is 16.00 g/mol. The molecular weight of water can be calculated by adding together the atomic masses of the elements in it. Thus, the context and questions must be clear when discussing these terms! Always be careful when reading textbooks and problems to see if when "molecular mass" is mentioned they actually mean "molecular mass" or if they mean "molecular weight" instead.Īs a guide to get you used to this, if I am using a term with its original definition, it will be in green, if I am using it with its colloquial definition it will be in red. The Mole How are chemists able to measure things when everything is made up of different atoms and, to make this more difficult, different isotopes of those atoms? With moles, of course! Not with the small subterranean mammal (though they are cute), but the unit moles.Īs I mentioned before, atomic mass and atomic weight are often used interchangeably, so molar mass, molecular mass, AND molecular weight are also often used interchangeably. Lastly, we will look at the link between molar mass and boiling point.Then, we will explain the concept of molecular mass and explain the differences between it and molar mass.Next, we will explain the concepts of atomic mass and molar mass and the differences between the two.First, we will define a mole and explain why that unit is used.This article is about moles and molar mass.In this article, we will discuss how chemists measure and group the masses of atoms and molecules. However, we first need to know the average mass of each atom. We treat atoms the same way! To find out how many atoms are in a sample, we can relate the mass of the sample to the number of atoms. If you know the mass, you can multiply that by the desired quantity and add the candy to a scale until you reach that weight. This is a very long and tedious way to do business! Instead, you utilize the average mass of the candy. Let's say you work at a candy store and a customer asks for different amounts of candy (100, 250, 1000…), so you count out each order.
